Subperiosteal Implant Manufacturer for the US Market: Custom Designs for Complex Cases
When an oral surgeon faces a patient with severe maxillary atrophy, an unusable bone volume, or a history of failed endosteal implants, the treatment path often narrows to one option: a custom subperiosteal implant. What happens next depends entirely on the manufacturing partner behind that case.
Finding a truly specialized subperiosteal dental implant manufacturer — one capable of producing patient-specific frameworks at the engineering precision these cases demand — is not the same as sourcing a standard implant component. The anatomy is unique. The design is unique. The stakes are high. And the manufacturing process has to match all of it.
At Zygomatic Pterygoid Surgical Guides, subperiosteal implant manufacturing is not a secondary product line. It is the core of what we do, and it is why clinics and distributors across the United States and internationally choose us for the cases that most manufacturers cannot reliably support.
Why Subperiosteal Implant Manufacturing Demands True Specialization
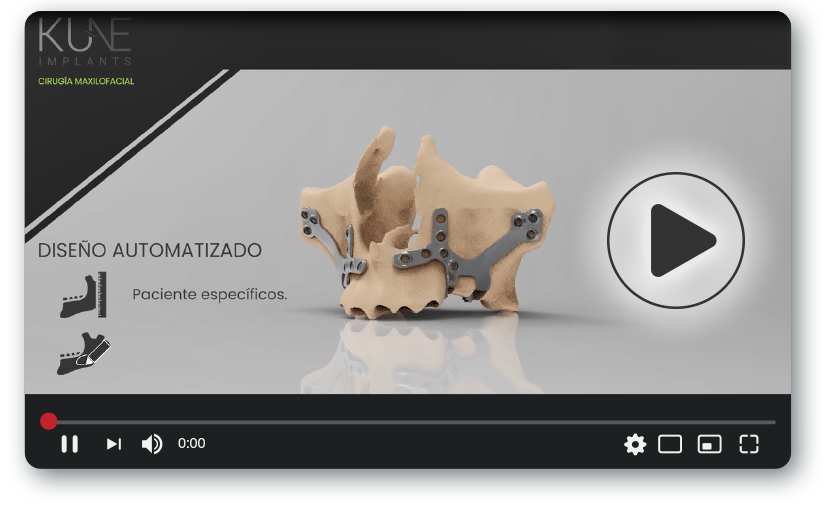
A subperiosteal implant sits on top of the bone, beneath the periosteum, rather than being placed within it. This fundamental difference means the implant cannot follow a standard geometry. It must conform precisely to the individual topography of each patient’s jaw — and that topography is never the same twice.
In patients with severe bone atrophy, compromised ridges, or anatomical irregularities from prior surgeries, this level of customization is not a premium option. It is a clinical requirement. A framework that does not achieve a true passive fit will generate micromovement, create soft-tissue complications, and ultimately compromise the prosthetic outcome.
This is why subperiosteal implant manufacturing demands far more than a machining facility. It requires a team that understands the anatomy, can interpret CBCT data with clinical accuracy, has the engineering capability to translate that anatomy into a precisely fitted framework, and knows how fit, fixation, and prosthetic alignment interact in the context of complex full-arch rehabilitation.
Generalist implant manufacturers — companies that produce hundreds of different implant types across a broad catalog — rarely build this depth in one specific area. The surgical and engineering disciplines required for consistent subperiosteal manufacturing are too specialized to be a side product.
What Distinguishes a Specialized Subperiosteal Implant Manufacturer from a General Dental Supplier
The gap between a generalist dental implant supplier and a dedicated subperiosteal implant manufacturer becomes most visible when a case gets complicated.
A generalist may offer a subperiosteal option, but what that typically means is a limited set of pre-designed frameworks adapted to approximate anatomy — not true bespoke manufacturing. When the patient’s anatomy falls outside the expected range, the process breaks down. The surgical team is left to adapt intraoperatively, and the implant’s performance reflects those compromises.
A specialized manufacturer starts from the patient’s actual anatomy and works outward. Every decision — from framework geometry to bar design, from screw positioning to emergence profile — is made in reference to that specific patient’s bone surface, nerve positions, and prosthetic needs.
Engineering Depth and Patient-Specific Design
At the foundation of true custom subperiosteal implant production is a digital planning workflow built around the patient’s CBCT scan data. This data becomes the basis for an anatomical model that engineers and clinicians review together before any manufacturing begins.
The patient-specific design process addresses not just the bone surface, but the relationship between the implant framework and the intended prosthetic restoration. Bar angulation, abutment positioning, and emergence geometry are planned as part of a unified restorative and surgical workflow — not as separate steps that are reconciled after the fact.
This level of engineering collaboration is what makes the difference between a subperiosteal implant that performs predictably over time and one that requires repeated clinical management.
Production Standards and Quality in Medical-Grade Manufacturing
Every subperiosteal framework produced at ZPSG is manufactured to medical-grade standards using precision CAD/CAM fabrication with materials appropriate for long-term intraoral performance. Each implant undergoes a rigorous quality review prior to dispatch — checking fit accuracy, structural integrity, surface quality, and design compliance against the approved clinical plan.
Passive fit is non-negotiable. The framework must seat fully and without tension the first time it is placed. This is not achievable without both a high-precision manufacturing process and strict quality control applied to every individual case.
Serving US Dental Clinics and Surgical Teams with Complex Case Capability
For dental clinics and surgical practices in the United States, working with an internationally specialized subperiosteal dental implant manufacturer means gaining access to engineering and manufacturing capabilities that simply do not exist at a local or general-purpose production level.
US-based oral surgeons and maxillofacial surgeons managing complex full-arch rehabilitation cases — particularly patients who have been declined for endosteal treatment due to insufficient bone volume — need a manufacturing partner they can trust to deliver a precise, well-planned, surgically viable implant on a reliable timeline.
That is the relationship ZPSG is built to provide.
From CBCT Scan to Delivered Framework: The Clinical Workflow
Working with our team follows a clear, structured process:
- Case submission — The clinical team submits the patient’s CBCT scan data along with relevant clinical information, prosthetic goals, and any specific anatomical notes.
- Engineering review — Our team analyzes the scan data, confirms the anatomical model, and opens a collaborative design dialogue with the submitting surgeon.
- Design proposal — A detailed framework design is prepared and shared for clinical review and approval. Adjustments are made iteratively before manufacturing begins.
- Fabrication — The approved design is manufactured to medical-grade standards using precision digital fabrication processes.
- Quality check — Each case is reviewed for fit accuracy, structural compliance, and surface quality.
- Delivery — The completed framework is dispatched with full documentation for surgical and prosthetic reference.
This workflow is designed to give surgical teams full visibility and clinical control at every stage — from the initial design decision through to the final delivered product.
Addressing Severe Atrophy, Revision Cases, and Anatomically Complex Patients
The cases that benefit most from bespoke subperiosteal implant manufacturing are also the ones that are hardest to treat by any other means.
Patients with severely resorbed maxillary or mandibular ridges, those with a history of multiple failed endosteal placements, individuals with unusual anatomical presentations from prior oncological treatment or congenital conditions — these are the patients for whom a custom subperiosteal framework is not an experimental option but a clinically rational one.
The ability to design around existing anatomy, to work with what the patient actually has rather than what the standard protocol requires, is what makes patient-specific subperiosteal manufacturing so valuable in these clinical scenarios. Every aspect of the framework is adapted to serve the patient in front of the surgeon — not an idealized average.
Global Subperiosteal Implant Distribution: Partnering Beyond the US
While our commercial focus includes strong engagement with the United States market, ZPSG operates as a global subperiosteal implant manufacturer and supplier. Clinics, surgical centers, and dental laboratories in multiple countries work with our team to access custom subperiosteal manufacturing for their most demanding cases.
We also collaborate with distributors who want to bring a specialized, high-quality subperiosteal implant solution to their regional professional networks. Distributor partnerships are a meaningful part of how we extend our manufacturing capabilities to markets where local access to this level of specialization does not yet exist.
If you represent a distribution network or regional dental supplier looking to offer advanced custom subperiosteal implants to your clinical partners, we welcome the conversation. You can learn more about our distributor collaboration model on our distributors page.
Why Clinics and Distributors Choose a Dedicated Subperiosteal Implant Partner
The decision to work consistently with one specialized subperiosteal implant manufacturer rather than sourcing from multiple generalist suppliers comes down to a few practical realities.
Predictability. When you work with a team that does only this, you know what to expect — in terms of design quality, production turnaround, communication responsiveness, and the reliability of the final product.
Technical depth. Complex cases require a manufacturing partner who understands the clinical context. Being able to have a real engineering dialogue about a difficult patient’s anatomy — rather than submitting a form and hoping the output is close — makes a measurable difference in clinical outcomes.
Efficiency. A well-structured submission-to-delivery process reduces back-and-forth, minimizes revisions, and gives surgical teams the certainty they need to plan procedures with confidence.
Long-term relationship value. The clinics and distributors that get the most from our manufacturing capability are the ones who build a working relationship over time — where the engineering team understands each practice’s clinical preferences and the collaboration becomes faster and more refined with every case.
A Manufacturing Partner That Understands the Case, Not Just the Component
The most technically precise subperiosteal implant is only as good as the clinical understanding that went into designing it. That is the principle that shapes everything ZPSG builds.
We are not a general-purpose implant supplier that added subperiosteal to a catalog. We are a dedicated manufacturing and planning partner for the cases where precision, customization, and genuine engineering collaboration are what determine whether a patient gets a successful rehabilitation or another clinical setback.
For US-based surgical teams and international clinics managing complex, high-stakes implant cases, we offer the manufacturing depth, the custom design workflow, and the case-by-case support that these patients require.
If you are working on a complex subperiosteal case, planning to expand your clinical offering, or exploring a distribution partnership, contact our team to start the conversation.

If you’re looking for a reliable partner for challenging “low-bone” or revision cases, contact us. We are a subperiosteal dental implant manufacturer specialized in custom & bespoke, patient-specific solutions, combining digital planning, precision titanium manufacturing, and strict quality controls to support predictable outcomes. Share your CBCT and case details, and our team will help you move from scan to a clinically driven design—built to fit, built to last.