Severely atrophic jaws, compromised ridge anatomy, post-traumatic defects, and failed grafting histories can make conventional endosteal implants unpredictable—or simply not feasible. In these scenarios, partnering with a subperiosteal dental implant manufacturer that delivers custom & bespoke subperiosteal implants can unlock a reliable path to full-arch rehabilitation without the long timelines and morbidity often associated with extensive bone augmentation.
Modern subperiosteal implant systems are not the “old school” designs many clinicians remember. Today, they are engineered through a digital workflow that combines CBCT data, CAD planning, and precision manufacturing—most commonly in medical-grade titanium—resulting in patient-specific frameworks designed to seat on the bone under the periosteum. For US-focused practices and dental labs, this approach is increasingly relevant for complex cases where patients demand fixed teeth solutions but present with advanced bone loss or anatomical constraints.
This article explains what a subperiosteal dental implant manufacturer in the USA market should provide, how custom devices are planned and fabricated, and which factors matter most when selecting a partner for high-risk, high-value cases.
What a US-Focused Subperiosteal Dental Implant Manufacturer Should Deliver
Choosing a manufacturer for patient-specific subperiosteal dental implants is not the same as choosing a commodity implant supplier. The right partner contributes to clinical success by supporting planning, optimizing design for soft-tissue management, ensuring accurate fit, and providing documentation and quality controls aligned with US expectations.
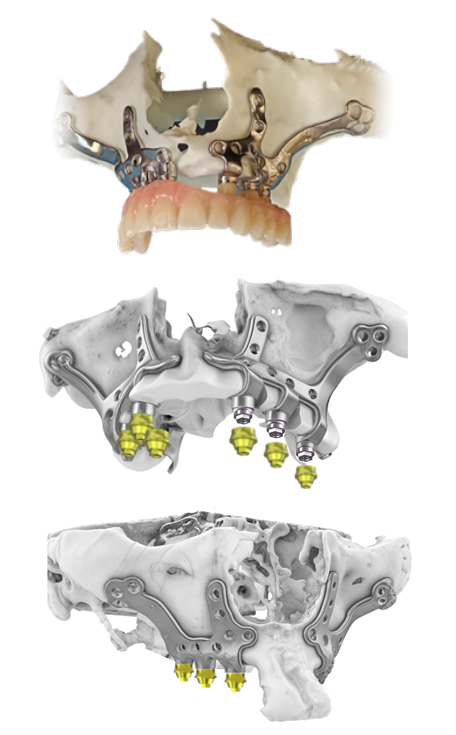
Digital Customization: Anatomy-Driven Design for “No-Bone” Cases
Subperiosteal implants are designed to adapt to the patient’s existing bone topography rather than requiring ideal bone volume for intraosseous anchorage. That single difference changes the engineering priorities and makes true customization essential. A capable custom subperiosteal implant manufacturer typically supports:
- CBCT/CT-based segmentation to reconstruct a precise 3D model of the maxilla or mandible
- CAD design of the subperiosteal framework to maximize stability, distribute load, and reduce pressure points
- Prosthetic-driven planning to align abutment positions with the final restoration, not just bone availability
- Consideration of critical anatomy (sinus floor, nasal cavity, nerve pathways, thin cortical plates, undercuts)
For complex cases—especially severe maxillary atrophy—custom design is the difference between a framework that seats passively and one that creates tension, irritation, or instability. “Bespoke” should mean the device is engineered around the patient’s anatomy and the restorative plan, not a lightly modified template.
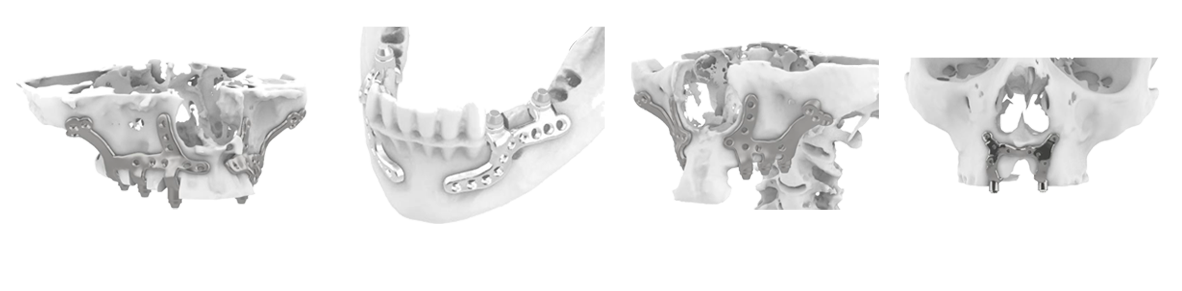
A Predictable Digital Workflow From Scan to Sterile Delivery
US clinics and labs typically expect a repeatable, auditable process. While workflows vary, most high-performing subperiosteal programs follow a structure like this:
- Case intake: CBCT (DICOM) plus intraoral scan (STL/PLY) or model scan when available
- Data validation: checking field of view, artifacts, slice thickness, and bite/occlusion records
- 3D reconstruction: segmentation of the jaw and identification of anatomical landmarks
- Design proposal: framework geometry, fixation strategy, and abutment emergence designed for restorative goals
- Clinical review and approval: iterative refinement with the surgeon/restorative clinician
- Manufacturing: precision production (commonly titanium) and finishing per protocol
- Quality checks: dimensional verification, surface inspection, cleaning
- Packaging and sterilization pathway: sterile delivery or validated sterilization instructions with documentation
A manufacturer that excels in complex cases does more than “make the part.” They help prevent avoidable complications by catching design issues early—before the framework ever reaches the OR.
Manufacturing Standards, Materials, and QA: What Matters for the US Market
If the goal is to rank and convert in the United States, your content should reflect what US clinicians care about: consistency, documentation, and support. A strong subperiosteal dental implant manufacturer should be able to speak clearly about:
- Medical-grade titanium selection and traceability
- Surface finishing that supports soft-tissue tolerance and long-term maintainability
- Dimensional accuracy and fit verification (passive seating is a clinical priority)
- Cleaning and packaging protocols to minimize residues and contamination risk
- Case documentation (revision history, approvals, manufacturing records)
In complex implant dentistry, the “details” are not minor. Pressure points, sharp transitions, and poorly planned abutment emergence can compromise soft tissue, hygiene access, and patient comfort—especially in full-arch cases where maintenance is critical.
Clinical Value: Why Custom Subperiosteal Implants Can Be the Right Tool
Subperiosteal solutions are not a universal replacement for endosteal implants. They are an option for specific indications, often when patients want fixed teeth but bone limitations restrict traditional approaches. Benefits commonly sought in complex subperiosteal implant cases include:
- Reduced need for extensive grafting and fewer staged surgeries in selected patients
- A more direct path to fixed rehabilitation, depending on clinical protocol and case selection
- Improved feasibility in severe atrophy where endosteal anchorage is compromised
- Prosthetic-driven outcomes, with abutments positioned to support restorative design
In practice, many US clinicians consider custom subperiosteal implants when patients cannot tolerate long reconstructive timelines, have failed previous grafting/implants, or present anatomy that makes conventional planning high-risk.
What to Look For When Selecting a Custom & Bespoke Manufacturer
From a US positioning standpoint, buyers want competence, responsiveness, and risk reduction. When evaluating a subperiosteal dental implant manufacturer, prioritize:
- Demonstrated subperiosteal experience (not only general 3D printing or milling)
- Engineer-to-clinician collaboration with clear revision cycles and design rationale
- Prosthetic planning capabilities (restoration-first design, hygiene accessibility, emergence profiles)
- Fixation strategy expertise (screw placement planning, stability without over-compressing tissues)
- Transparent turnaround and case communication (predictable timelines, clear deliverables)
- Documentation and traceability aligned with professional expectations in the US
- Post-delivery support for surgical questions, restorative alignment, and troubleshooting
A reliable partner will proactively address common failure modes: unstable seating due to scan artifacts, abutments that complicate prosthetic access, edges that irritate soft tissue, or frameworks that concentrate stress in thin bone.
Complex Case Scenarios Where Bespoke Subperiosteal Solutions Are Often Considered
Every case requires clinical judgment, but custom subperiosteal implants are frequently explored in scenarios such as:
- Severe maxillary atrophy where sinus augmentation or zygomatic options are not preferred
- Atrophic mandible with limited vertical bone and heightened risk profiles
- Revision cases after failed implants, peri-implantitis, or compromised graft sites
- Post-traumatic or post-resection defects requiring patient-specific adaptation
- Patients seeking fixed teeth who decline multi-stage grafting or prolonged treatment timelines
For these patients, the ability to engineer a patient-specific framework—rather than forcing anatomy into a standard implant pathway—can be clinically valuable and commercially differentiating for US practices.

If you’re looking for a reliable partner for challenging “low-bone” or revision cases, contact us. We are a subperiosteal dental implant manufacturer specialized in custom & bespoke, patient-specific solutions, combining digital planning, precision titanium manufacturing, and strict quality controls to support predictable outcomes. Share your CBCT and case details, and our team will help you move from scan to a clinically driven design—built to fit, built to last.

